Eczema treatment: Crisaborole (Eucrisa®) ointment
Crisaborole ointment
A newer treatment for eczema/atopic dermatitis, crisaborole ointment offers an alternative to treating eczema with corticosteroids that you apply to the skin, pimecrolimus cream, or tacrolimus ointment.

Why do dermatologists prescribe this medication to treat eczema/atopic dermatitis?
This is a newer treatment for people who have mild to moderate eczema. Studies show that crisaborole ointment can effectively treat:
Inflammation (redness, swelling, heat)
Itch (shown to relieve the itch in a few days)
Rash
FDA approval
The U.S. Food and Drug Administration (FDA) has approved crisaborole ointment to treat people who:
Have mild to moderate eczema
Are 3 months of age or older
Safety and effectiveness
In the clinical trials that led the FDA to approve crisaborole, the researchers enrolled 1,522 patients who had mild to moderate eczema. Their ages ranged from 2 to 79 years old.
To test the safety and effectiveness of this medication, some patients received crisaborole ointment. The other patients were given a moisturizing ointment that looked like crisaborole, but the ointment contained no medication.
In these clinical trials:
About one-third of the participants treated with the crisaborole ointment had clear or almost clear skin after applying the ointment twice a day for 28 days.
Crisaborole was found to relieve the itch in more patients than did the moisturizer. On day 2 of the study, 34% of patients using crisaborole had stopped itching vs. 27% using the moisturizer. By the end of the study, 57% of the patients treated with crisaborole no longer had itchy skin vs. 40% using the moisturizer.
The most common side effect of crisaborole is burning or stinging where you apply it. This usually happens immediately after applying crisaborole.
Less than 2% of patients experienced side effects that caused them to stop using the medication. Just as many patients who received the moisturizer stopped using it due to side effects.
In a longer study that ran 48 weeks, researchers found that few patients developed side effects.
More research is needed to learn the long-term effects.
How to use crisaborole
Use as directed
You apply a thin layer of crisaborole ointment to the skin as directed. It’s usually applied twice a day.
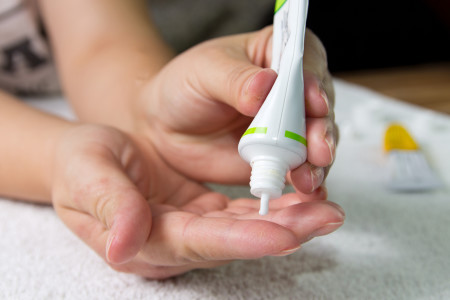
Best results are obtained when you use this medication along with:
Bathing
Eczema friendly moisturizer
Trigger management
Apply after bathing: Dermatologists recommend applying crisaborole immediately after getting out of the water. After applying crisaborole, wait 15 minutes before applying your moisturizer.
Wash your hands after applying crisaborole: There is one exception to this advice. If you are treating your own hands with this medication, do not wash your hands after applying it.
If you are not treating your hands or you are applying crisaborole to someone else, wash your hands immediately after you apply this medication.
Possible side effects
The most common side effects reported during the clinical trials were:
Burning or stinging when applied to the skin
Worsening eczema
Skin infection
Fewer than 4% of patients experienced any one of these side effects.
Tip: If crisaborole stings when you apply it, keep this medication in the refrigerator. Applying it cold can prevent that stinging sensation.
When should I call my dermatologist?
Call your dermatologist, if you apply crisaborole and notice any of the following:
Eczema worsens
Skin infection develops
No improvement after applying for 4 weeks
Related AAD resources
Images
Getty Images
References
Eichenfield LF, Call RS, et al. “Long-term safety of crisaborole ointment 2% in children and adults with mild to moderate atopic dermatitis.” J Am Acad Dermatol 2017;77:641-49.
Paller, A, Tom WL, et al. “Efficacy and safety of crisaborole ointment, a novel, nonsteroidal phosphodiesterase 4 (PDE4) inhibitor for the topical treatment of atopic dermatitis (AD) in children and adults.” J Am Acad Dermatol 2016;75:494-503.
Yosipovitch G, Gold LF, et al. “Early relief of pruritus in atopic dermatitis with crisaborole ointment, a non-steroidal, phosphodiesterase 4 Inhibitor.” Acta Derm Venereol. 2018;98(5):484-9.
Last updated: 1/31/23
 Atopic dermatitis: More FDA-approved treatments
Atopic dermatitis: More FDA-approved treatments
 Biosimilars: 14 FAQs
Biosimilars: 14 FAQs
 How to trim your nails
How to trim your nails
 Relieve uncontrollably itchy skin
Relieve uncontrollably itchy skin
 Fade dark spots
Fade dark spots
 Untreatable razor bumps or acne?
Untreatable razor bumps or acne?
 Tattoo removal
Tattoo removal
 Scar treatment
Scar treatment
 Free materials to help raise skin cancer awareness
Free materials to help raise skin cancer awareness
 Dermatologist-approved lesson plans, activities you can use
Dermatologist-approved lesson plans, activities you can use
 Find a Dermatologist
Find a Dermatologist
 What is a dermatologist?
What is a dermatologist?